
Eric Bangeman / Ars Technica
The usa is in the end beginning to check for the coronavirus in vital volumes. On Thursday, the overall collection of coronavirus exams carried out in The usa crowned 100,000, in keeping with the COVID monitoring mission. That is a 10-fold build up from every week previous. Within the coming days, we will be able to be expecting the tempo of trying out to proceed expanding as an increasing number of organizations—each instructional labs and for-profit corporations—ramp up trying out efforts.
That is essential as a result of The usa has a large number of catching as much as do. A sequence of early missteps on the federal degree hampered The usa’s trying out efforts within the early weeks of the coronavirus disaster. It wasn’t till the top of February that the Meals and Drug Management opened the door for a variety of organizations to provide coronavirus exams.
Within the remaining week, we now have began to look the result of that modify. Personal corporations are beginning to send loads of hundreds of exams. Pressure-through trying out stations are sprouting up everywhere The usa.
However the explosion of recent trying out efforts will create demanding situations of its personal. Josh Sharfstein, a professor on the Johns Hopkins College Faculty of Public Well being, argues that coordination might be an increasing number of essential as the quantity of trying out will increase. A Thursday tale in The Wall Boulevard Magazine painted an image of chaos as sufferers struggled to get examined—even after they have gotten a referral from a health care provider to get a check. Some drive-through trying out amenities have had lengthy traces and unpredictable hours, the Magazine stories.
To get most worth out of all this trying out, officers are going to want to rationalize and streamline those trying out processes. Sufferers want predictable laws for when they’re eligible to be examined and a predictable procedure for if truth be told getting a check and getting effects. And public well being officers want standardized tactics of accumulating check effects
Some labs also are affected by a scarcity of provides, Axios stories. As we detailed in our February explainer, trying out for the virus at the back of COVID-19 is a multi-step procedure that comes to plenty of chemical reagents. Those chemical substances are most commonly to be had off-the-shelf, however the machine is not designed to maintain unexpected, huge spikes in call for.
“We’re holding redundancy very a lot at the again of our minds,” Harvard’s Michael Mina informed Axios. “There’s a worry that some merchandise may develop into out of inventory and in order quickly as we’re reside with our check, we can get started validating different changes.”
We noticed this first-hand. On Thursday, Ars Managing Editor Eric Bangeman drove to a drive-through trying out website close to his house within the Chicago space to get some footage for this tale. On Friday, the health center web hosting the ones exams introduced it used to be postponing its drive-through trying out program because of a scarcity of check kits.
Nonetheless, there is each explanation why to be expecting that trying out suppliers will determine those kinks. The collection of coronavirus exams soared this week, and we will have to be expecting that growth to proceed within the coming weeks. And that’s the reason essential as a result of The usa goes to do much more trying out to get the coronavirus outbreak beneath keep an eye on.
Federal missteps intended The usa began out at the back of on trying out
The usa began out at the back of on trying out capability because of a chain of early errors by way of federal companies. The tale begins on January 31, when the Secretary of Well being and Human Services and products declared the radical coronavirus to be a public well being emergency. Because of a quirk in federal legislation, this declaration intended that any one in need of to check for the coronavirus first had to search approval from the Meals and Drug Management.
The FDA has a large number of discretion in the way it makes use of that authority. The company can have arrange a streamlined procedure to permit all kinds of organizations to create their very own exams. Nevertheless it did not do this.
The Facilities for Illness Keep an eye on used to be operating on a coronavirus check, and to start with the FDA determined to not permit someone else to broaden exams of their very own. Sadly, when the CDC despatched out an early batch of coronavirus trying out kits to state and native laboratories, most of the labs had been not able to validate the effects.
The CDC’s efforts to troubleshoot this flaw wasted precious time.With a restricted collection of exams to be had, the CDC to start with restricted trying out to those that had just lately traveled to coronavirus hotspots or those that had come into touch with a recognized coronavirus service. That made it tough to trace early instances of neighborhood unfold inside the USA.
Coronavirus exams are rising exponentially

Eric Bangeman / Ars Technica
In any case, on February 29, the FDA opened the door to different labs growing their very own exams. Beneath the FDA’s new framework, labs that met sure prior regulatory necessities could be allowed to start out the usage of their very own exams ahead of they might gotten specific approval from the FDA. Beneath the brand new steering, labs had a 15-day grace length when they started trying out to record the vital bureaucracy with the FDA.
“That modified the entirety,” one lab director informed the New Yorker. “We didn’t have to attend to get the bureaucracy in after which stay up for the reaction.”
That call opened the floodgates. Within the remaining 3 weeks, the quantity of coronavirus trying out has grown exponentially. Knowledge from the COVID Monitoring Venture displays that the overall collection of coronavirus exams carried out in the USA grew from fewer than 1,000 on March four to just about 10,000 by way of March 12 and greater than 100,000 by way of March 19.
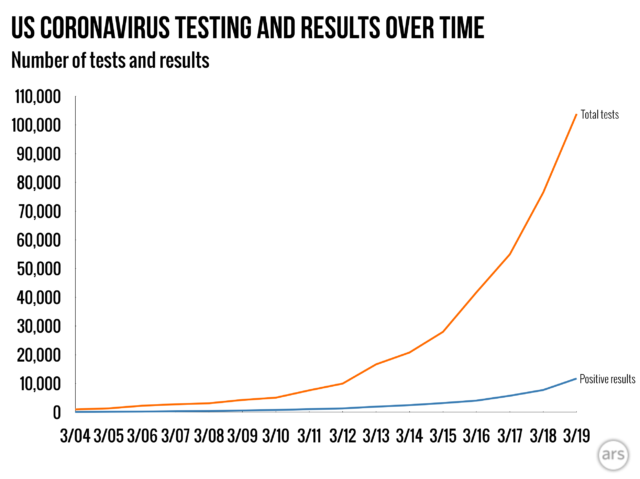
Those numbers are nonetheless some distance from enough to get the coronavirus beneath keep an eye on. South Korea, with about 15 p.c of the USA inhabitants, has examined 270,000 other people. However The usa’s trying out capability has been rising rapid, and we will be able to be expecting endured expansion within the coming days and weeks.
On Wednesday, Abbott Labs introduced FDA approval of its coronavirus check. The corporate mentioned it used to be “deploying 150,000 laboratory exams instantly” and used to be aiming to succeed in one million exams every week ahead of the top of March.
Any other main clinical trying out supplier, Roche, introduced on Monday that it used to be making plans to send out 400,000 exams. “The check kits are being despatched to a community of greater than 30 health center and reference laboratories in the USA that have already got the specified instrumentation in position and be capable to put in force high-volume trying out instantly.”
Those are simply two of many examples of US suppliers operating to ramp up coronavirus trying out capability. With their lend a hand, the USA will quickly have way more coronavirus trying out capability than it has nowadays.
Why trying out is essential
Checking out is not simplest essential as it is helping person sufferers perceive their clinical stipulations and information their remedy—despite the fact that that is clearly essential. In an endemic, trying out may be a very powerful to lend a hand public well being officers successfully direct assets, get the unfold of the virus beneath keep an eye on, and in the end allow other people to go back to customary lives.
Till just lately, well being care pros and public well being officers had been flying blind. They might simplest check a small fraction of the sufferers who exhibited coronavirus-like signs, and so they have tended to order exams for sufferers who exhibited critical, probably life-threatening signs. Different sufferers are normally informed to move house and self-isolate—with out figuring out needless to say if they’ve the coronavirus.
That uncertainty makes it onerous to take precautionary steps to stop additional unfold of the virus. If a affected person is in a position to ascertain that they’ve the coronavirus, then they (or public well being officers) can warn pals, circle of relatives, and different shut contacts in regards to the possible an infection. Preferably, the ones other people can get examined as neatly. However with no trying out, public well being officers do not know which instances to concentrate on.
And big-scale trying out will in the end permit public well being efforts to develop into extra centered, permitting extra American citizens to go back to customary lives. That would possibly not occur instantly, Johns Hopkins knowledgeable Josh Sharfstein informed Ars. There at the moment are such a lot of coronavirus instances that it’ll require further weeks of fashionable social distancing measures to deliver the outbreak beneath keep an eye on.
However as those measures begin to have an affect and an infection charges fall, trying out will permit public well being officers to exactly goal additional suppression efforts. Officers will be capable to establish spaces the place the coronavirus has been stamped out and other people can safely resume customary actions. They’re going to establish different spaces that stay coronavirus hotspots and require extra public well being assets. As infections develop into uncommon and trying out turns into considerable, officers will be capable to check shut contacts of newly stricken people, making it much less most likely that a unmarried new coronavirus case will change into a cluster of them.



